Analytical Instrumentation
|
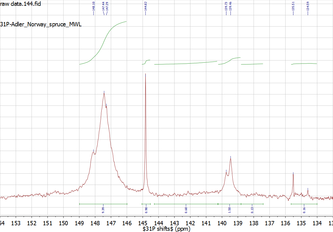
We use advanced quantitative 31P NMR for
- identifying and quantifying lignin termination motifs and lignin interunit bonding motifs carrying hydroxyl groups, - analyzing lower molecular weight polyphenols, such as tannins, - quantifying technical loading data for functionalised lignins. Samples are analyzed using NMR facilities at the Department of Chemical Sciences and Technologies at the University of Rome 'Tor Vergata'. |
|
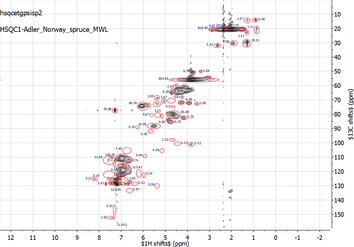
We use advanced qualitative and quantitative bi-dimensional NMR techniques such as {13C-1H}-HSQC for
- identifying and quantifying lignin interunit bonding motifs, - analyzing lignin-carbohydrate complexes (LCCs), - analyzing modified lignins and lignin-containing hybrid materials. Samples are analyzed using NMR facilities at the Department of Chemical Sciences and Technologies at the University of Rome 'Tor Vergata' or at the Large-Scale Facility for NMR Spectroscopy at the Bijvoet Center for Biomolecular Research at the Utrecht University. |
|
We use MALDI-TOF-based analysis for
- analyzing polymer composition of oligomeric tannins, - analyzing oligomeric lignin fractions for sequencing information - quantifying technical loading data for functionalised lignins. |
|
We use gel permeation chromatography with UV- and refractive index-based detectors for
- determining molecular weight key figures of natural polymers, - the development and evaluation of analysis protocols for natural polymers and oligomers. We use liquid chromatography with UV- and refractive index-based detectors for
- analyzing the purity of lower molecular weight natural polyphenols, such as tannins and low molecular weight lignin fractions, - the purification of of lower molecular weight natural polyphenols. |
|
We use liquid chromatography coupled with a UV-detector and an electron spray ionization (ESI)-based mass detector for
- reaction monitoring - dynamic release studies - determining low molecular weight (by-)products of biotechnological lignin transformations, - investigating mechanistic interactions of lower molecular weight polyphenols with small molecules. |
|
We use gas chromatography coupled with a flame ionization detector and/or an electron ionization (EI)-based mass detector for
- determining low molecular weight (by-)products of biotechnological lignin transformations, - investigating mechanistic interactions of lower molecular weight polyphenols with small molecules, - head-space analyses, - analysis of low MW extractives, - analyses in DOE-studies, - reaction controls, - static release studies. |
|
We use UV-vis spectroscopy for
- the determination of enzyme loadings, - the determination of enzyme activities, - the determination of enzyme kinetics, - the quantification of functional groups in natural polyphenols, - mechanistic evaluation of interactions involving polyphenols, - static release studies. |
|
We use scanning electron microscopy for
- analyzing fibrous nanomaterials based on natural polyphenols, - analyzing nanoparticles made from natural polyphenols. Samples are analyzed using the SEM facility at the Department of Chemical Sciences and Technologies at the University of Rome 'Tor Vergata'. |